
Exquisite crystalline snowflakes capture the imagination. But what is the science behind their formation, and is it true there are no two snowflakes alike?
First of all, the temperature and humidity of the atmosphere determine the shapes of snowflakes. And snowflakes form in the atmosphere when cold water droplets freeze onto dust particles. Finally, depending on the temperature and humidity of the air where the snowflakes form, the resulting ice crystals will grow into a myriad of different shapes.
Wilson Bentley
Wilson Bentley (1865–1931) from Jericho, Vermont, was the first person to capture photographs of snowflakes through a microscope attached to a camera. His collection of over 5,000 images – no two alike – introduced many people to the astounding diversity of snow crystals.


Classification systems
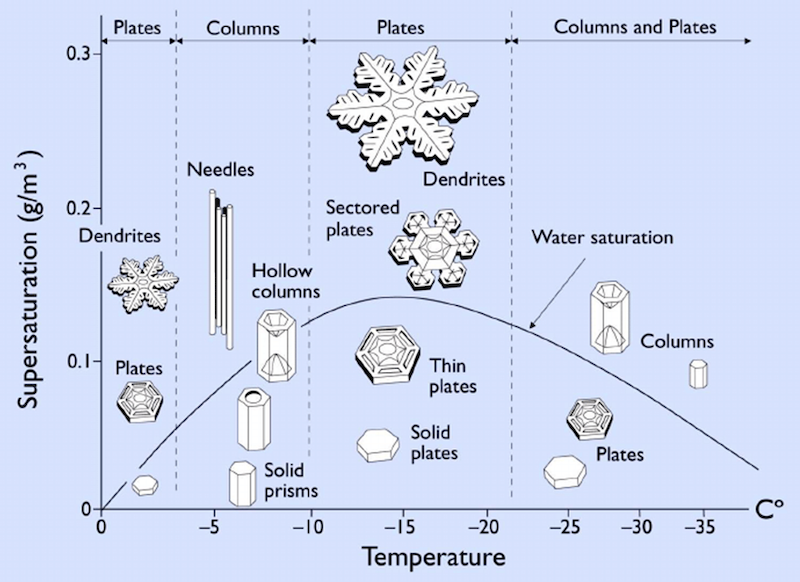
In 1951, scientists from an organization now called the International Association of Cryospheric Sciences (IACS) devised a classification system that characterized snowflakes into 10 basic shapes. Kenneth Libbrecht, professor of physics at the California Institute of Technology, in his guide to snowflakes at SnowCrystals.com, provides this chart of 35 types of snow crystals. They range from ice fragments to rods to the much more complex fern-like dendritic flakes we are all familiar with.

How snowflakes form from water vapor
Libbrecht has made extensive observations of how water molecules get incorporated into snow crystals. In his research, he observed that the most intricate snowflake patterns form when there is moisture in the air. Snowflakes produced in drier conditions tend to have simpler shapes.
According to Libbrecht’s research, temperature also has a large effect on the formation of snowflakes. Snowflakes formed in temperatures below -7.6 degrees Fahrenheit (-22 degrees C) consist primarily of simple crystal plates and columns. Meanwhile, snowflakes with extensive branching patterns are formed in warmer temperatures.
Visit Libbrecht’s website SnowCrystals.com for a wealth of information about snowflakes, including short videos of lab-grown snowflakes forming.

A gallery of snowflakes by Wilson Bentley




Bottom line: Temperature and humidity influence snowflake shape. The most intricate snowflake patterns form during warm and wet conditions.











