Astronomers have been arguing for some years about comets brought Earth its water. Then in 2011, an international team of astronomers using the Herschel Space Observatory to study Comet Hartley 2 (103P/Hartley) published their results on the first comet confirmed to contain ocean-like water.
Besides life, the biggest thing that distinguishes the Earth from other planets in the solar system is the presence of copious amounts of liquid water. Water molecules have been found in nebulae in distant reaches of the galaxy, so water itself isn’t uncommon in the universe. So it might be surprising to learn that no one really knows how all the water on Earth got here!

Over the years, four prominent theories explaining the origin of water on Earth have gained favor. In one, water-rich asteroids and meteorites impacted the infant Earth, distributing water across the planet by brute force. In another more serene process, the oceans formed when hydrogen and oxygen in the materials that made up the Earth (e.g., hydrocarbons and oxygen in iron oxides) combined chemically below the Earth’s crust and emerged as volcanic steam that condensed and rained down on the surface. A more recent theory suggests that water molecules actually adhered to the surfaces of the interstellar dust grains that accreted to form the solar system. In that case, water accumulated simultaneously with the rest of the planet. And last, but not least, there are the comets.
For decades, the accepted wisdom has been that comets brought a large proportion of water to the primordial Earth. In spite of the seemingly logical connection between comets and oceans, there has been one serious problem with that theory: the composition of water thus far detected in comets has differed fundamentally from that of the Earth’s oceans, so they couldn’t possible be a primary source. This problem was serious enough to threaten the comet source model altogether. Or at least it was until now.
Not all water is created equal
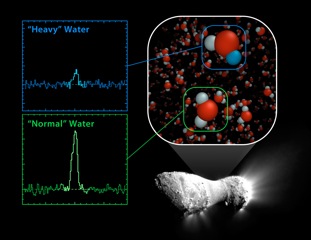
The composition problem that has dogged the comet model is rooted in the atomic structure of ocean water. It turns out that not all ocean water is made up of “regular” water (i.e., H2O). About one out of every 3,200 water molecules in the ocean is a heavy water molecule made with deuterium — a hydrogen atom with an extra neutron. When this hydrogen isotope combines with oxygen to make water, it’s actually about 10 percent heavier than the much more common form of water found everywhere around us on Earth.
Any theory of water transport to the Earth from space must account for this specific ratio of regular to heavy water molecules. This is why many researchers favor, for example, the asteroid impact model; scientists have verified that asteroids and some meteorites do contain the right ratio of heavy to regular water.
For comets to be a source of the Earth’s ocean water, they too must contain just the right ratio of heavy to regular water. But until Comet Hartley 2, no comet had been found to meet this vital criterion.
In fact, the specific chemistry of comets was unknown until the 1980s, when the first direct measurements of comet ice were made on Halley’s Comet and — years later — Comet Hyakutake. Unfortunately, these two comets contained twice as much heavy water than is found in water on Earth. That meant they, and comets like them, couldn’t possibly be a source of ocean water. The comet model was sinking, fast.
But scientists weren’t willing to give up. In 2000, scientists seized a rare opportunity to make another measurement of comet water when Comet LINEAR broke up as it approached the sun. While the right proportion of deuterium to hydrogen was not directly measured, other chemical tracers strongly suggested that deuterium was present in just the right amount required to explain ocean water composition.
For the next 10 years, the jury was still out on whether or not comets could contain the right amount of deuterium. Nowadays, thanks to Comet Hartley 2, it appears that comets are back in the game!
It is believed that comets like Hartley 2 and LINEAR, both of which originated in the Kuiper Belt near Jupiter’s orbit, possess the appropriate amount of heavy water. Finding such comets is challenging since, over time, gravitational perturbations have depleted that source of comets. Comets Halley and Hyukatake did not originate in the same region, which explains their completely different chemical compositions.
Ted Bergin of the University of Michigan – a member of the team that discovered ocean-like water in Comet Hartley 2 in 2011 – acknowledged that the result is based on a sample of one. He told EarthSky last fall:
We really need to know if this comet is a representative member of the Kuiper Belt. It is one very important measurement but we need more to begin to put the pieces of this puzzle together.
The results show that the amount of material out there that could have contributed to Earth’s oceans is perhaps larger than we thought. What this adds to the story is that the reservoir of material that can potentially be brought to the Earth with the right “kind” of water is much larger. This does not say that comets did bring water to the Earth but rather that they might.
While it is most likely that water came to Earth through a variety of processes, this latest finding reinvigorates the theory that comets may have contributed a lot more water to the Earth than was recently thought.
Now, as for the origin of comets themselves? That’s a question for another rainy day.
Bottom line: Astronomers have been arguing for decades about how Earth got its water. In 2011, using the Herschel Space Observatory to study comet Hartley 2 (103P/Hartley), an international team of astronomers including Ted Bergin of the University of Michigan found the first comet confirmed to contain ocean-like water. The comet is Comet Hartley 2. These results appeared online October 5, 2011, in the journal Nature.
Icy heart of Comet Hartley 2 is tumbling, at a changing rate











